
Also, you have a built in answer key in the form of the periodic table. Because there are way more carbon-12 atoms than carbon-13 atoms in nature (98.90% versus 1.10%) we would expect the average atomic mass is closer to 12 amu than to 13 amu. There are a few ways we can check answers and avoid mistakes on questions that involve calculating average atomic masses. Remember to convert the percentages into fractions before doing the calculation or you'll end up with some outrageously incorrect atomic mass. Check this out:Īverage atomic mass of carbon = (percent abundance 12C)(atomic mass 12C) + (percent abundance 13C)(atomic mass 13C)Īverage atomic mass of carbon = (0.9890)(12.000 amu) + (0.0110)(12.00335 amu) The average atomic mass can be calculated by multiplying the percent abundance by the atomic mass for each isotope then adding the values together. We can calculate the average atomic mass of carbon by using the weight of each isotope and the percent natural abundance of each isotope.Ĭheck out this data for the carbon isotopes 1: Not to be confused with the atomic number ( Z), which is the number of protons. The number in superscript or after the dash indicates the mass number ( A) of the atom which is the number of protons and neutrons. While we're at it, you may also see carbon-12 written as 12C or carbon-13 written as 13C. 1 We say naturally occurring because other isotopes of carbon can be made in the lab. The periodic table shows the average mass of the mixture of isotopes for each element.Ĭarbon, for example, has two naturally occurring isotopes, carbon-12 and carbon-13. Isotopes are atoms of the same element that contain different number of neutrons in their nuclei. The difference is because most elements (including carbon) have more than one isotope. 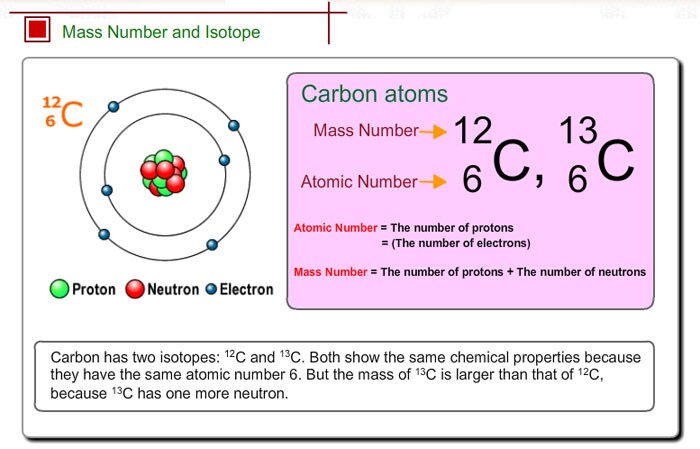
What gives? Fear not, sleuth-there is a reasonable explanation. Wait a minute, Shmoop, that's not 12.00 amu as we defined it earlier. Perhaps you're a regular Sherlock Holmes and you notice the atomic mass given for carbon (C) on the periodic table above is 12.01 amu. The periodic table: the atomic masses are located under the elemental symbols. That's where all the atomic masses on the periodic table come from. Although we do not know how much the average iron atom's mass is, we do know that it is 56 times as massive as a hydrogen atom. In other words, it not necessary to put a tiny atom on a scale to weigh it as long as we have a standard we can compare it to. If it's determined experimentally you weigh relatively 22% of what The Hulk weighs, you would weigh 0.22 HUs. 
We'll say The Hulk weighs exactly one Hulk unit (HU). Let's say we all compare ourselves to The Hulk. To fix this predicament, we could create a new scale system that compares our weight relative to others. Let's say all of the scales in the world are busted. That means the atomic mass of a hydrogen atom is 8.4% of that of a carbon-12 atom (1.008 amu to be exact). For example, the hydrogen atom is 8.4% as massive as the carbon-12 atom. How can this information help us determine the atomic masses of other elements? Experiments can be done to determine the relative mass of an element compared to carbon. The atomic mass is defined as the mass of an atom expressed in atomic mass units. Who comes up with this stuff? The carbon-12 atom is the atom that has six protons and six neutrons. One atomic mass unit (amu) is defined as a mass equal to one-twelfth the mass of a single carbon-12 atom. The first step is to assign a mass value to a chosen element to act as a standard. We can determine the mass of one atom relative to another atom. (Although we heard the Smurfs are hiding one.) That's not going to stop us, though. But how do we determine the weight or mass of an atom? It's not as if we have a scale small enough to measure such a small weight. The mass of an atom is determined by the number of protons, neutrons, and electrons it contains. Stoichiometry will seem easy peasy lemon squeezy once we're finished. These relationships will help us study the composition of compounds and how that composition can change during a reaction. It's more like the relationship between members in a group of friends-and the friends are different types of elements. We're not talking about the "it's complicated" type of relationship. We're going to use them to study the relationship between the mass of atoms and the mass of molecules. Remember chemical structures and formulas? Yeah, they're still important.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
